
来源:admin 浏览量: 发布时间:2022-04-19 10:16:27
来源:https://mp.weixin.qq.com/s/moTK2MSfGrgPJh55HLo6fw
 第一作者:魏凯
第一作者:魏凯
通讯作者:艾智慧教授(华中师范大学)、张礼知教授(华中师范大学、上海交通大学)
论文DOI: 10.1016/j.watres.2022.118453
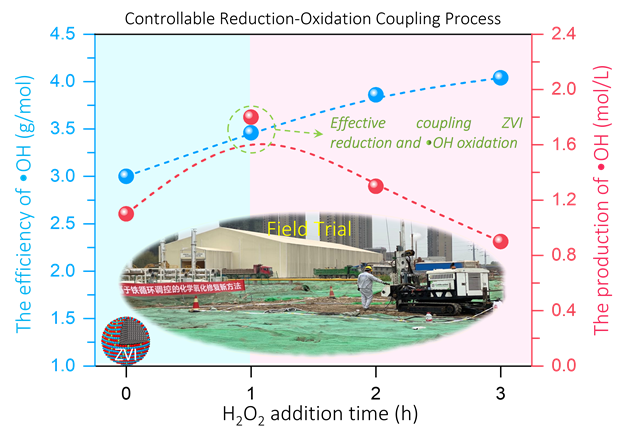
构建ZVI/H2O2 ROC过程
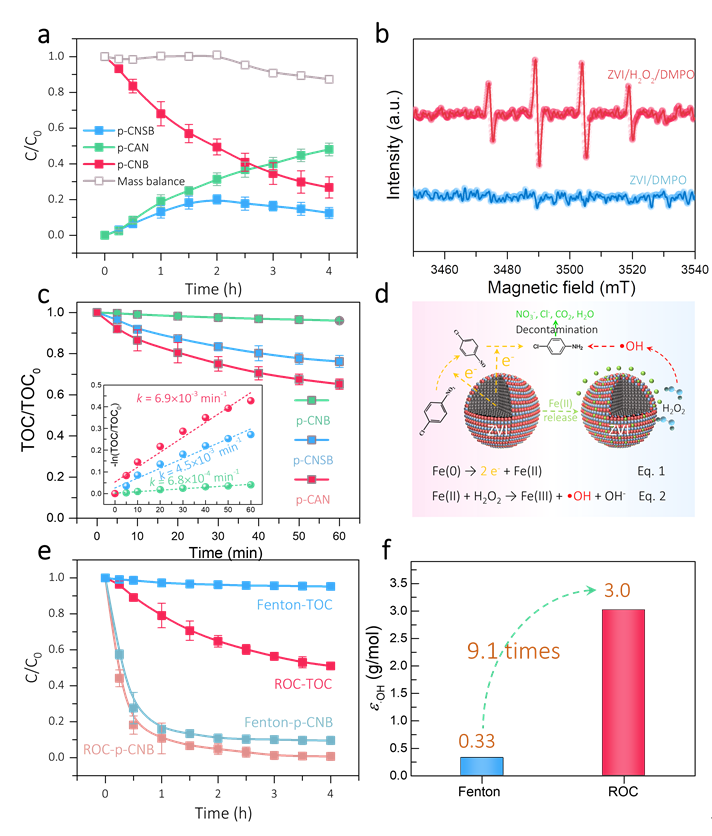
Figure 1. Construction of ZVI/H2O2 based ROC process. (a) Time profiles of p-CNB reduction by ZVI, reduction mass balance = [p-CNB] + [p-CNSB]+ [p-CAN]. (b) The EPR spectra of ZVI and ZVI/H2O2 processes with DMPO as spintrapping agent. (c) Time profiles of the TOC removal of p-CNB, p-CNSB and p-CANin Fenton oxidation process. (d) Schematic illustration of the possible ROC process constructed by ZVI and H2O2 for p-CNB treatment. The black, red, blue, and green balls represented Fe(0), Fe(III), oxygen, and Fe(II), respectively. (e) Time profiles of p-CNB degradation and corresponding TOC removal in ROC, and Fenton oxidation processes. (f) The removed amount of TOC per-molar •OH toward p-CNB in ROC and Fenton oxidation processes. The initial concentrations of p-CNB, p-CNSB, and p-CAN were 0.1 mmol/L. The dosages of ZVI, Fe(II), and H2O2 were 0.8 g/L, 14.3 mmol/L, and 5 mmol/L, respectively, initial pH was 3.0. Copyright 2022, Elsevier Inc.
ZVI可以还原对氯硝基苯(p-CNB)为对氯亚硝基苯(p-CNSB)和对氯苯胺(p-CAN),从而增强氧化过程中•OH氧化矿化污染物的效率。ZVI/H2O2 ROC过程中的ε•OH为3.0 g/mol,是单独氧化过程(Fenton)的9.1倍(0.33 g/mol),证实ZVI还原可以提高•OH氧化矿化污染物的效率。
ZVI/H2O2 ROC过程的可控性

Figure 2. Controllability of ZVI/H2O2 based ROC process. (a) Schematic illustration of the controllable ZVI/H2O2 based ROC processes by tuning the addition time of H2O2. The black, red, blue, and green balls represented Fe(0), Fe(III), oxygen, and Fe(II), respectively. (b) Time profiles of the mineralization of p-CNB in different ROC processes.(c) Time profiles of accumulative •OH production in different ROC processes. The initial concentrations of p-CNB, ZVI, and H2O2 were 0.1 mmol/L, 0.8 g/L, and 5 mmol/L, respectively, initial pH was 3.0. Copyright 2022, Elsevier Inc.
通过控制ZVI/H2O2 ROC过程中H2O2的添加时间,可以调控p-CNB的还原程度和•OH的生成量,从而控制氧化矿化p-CNB的表现。
ZVI/H2O2ROC过程的可控机理

Figure 3. Fe(II) concentration variation in ZVI pre-reduction process. The SEM images of ZVI with the reduction time of (a) 0 h, (b) 1 h, (c) 2 h, and (d) 3 h. HR-XPS of Fe 2p ofZVI with the reduction time of (e) 0 h, (f) 1 h, (g) 2 h, and (h) 3 h. The initial concentrations of p-CNB and ZVI were 0.1 mmol/L and 0.8 g/L, initial pHwas 3.0. (i) Time profiles of surface and dissolved Fe(II) concentrations in ZVI/H2O process. The initial concentration of ZVI was 0.8 g/L, initial pH was 3.0. (j) Schematic illustration of the temporal Fe(II) concentration in ZVI pre-reduction process. Copyright 2022, Elsevier Inc.
不同预还原时间对应着不同的ZVI腐蚀程度。相应地,ZVI表面和溶出Fe(II)的浓度也会随着还原时间延长而改变。其中,预还原1 h时,ZVI表面和溶出Fe(II)的浓度均达到最大值。此时添加H2O2,体系具有最高的H2O2分解效率,生成最多的•OH,可以最大化地耦合还原和氧化过程。
p-CNB矿化路径
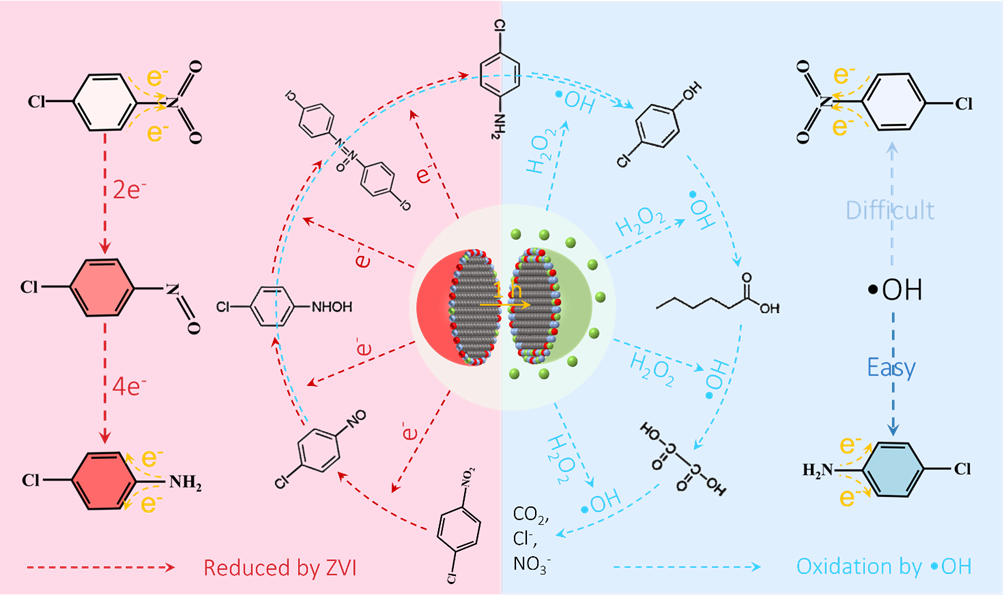
Scheme 1. Proposed p-CNB mineralization pathway in the ROC-1 process. The black, red, blue, and green balls represented Fe(0), Fe(III), oxygen, and Fe(II), respectively. Copyright 2022, Elsevier Inc.
气相色谱-质谱和离子色谱结果揭示ZVI可以逐步地还原p-CNB为p-CNSB、对氯苯羟胺、对氯氧化偶氮苯、p-CAN等,在此过程中,吸电子的硝基逐渐转化为供电子的氨基,导致苯环上电子云密度增加,有利于随后氧化过程中苯环氧化开环,最终实现完全矿化。
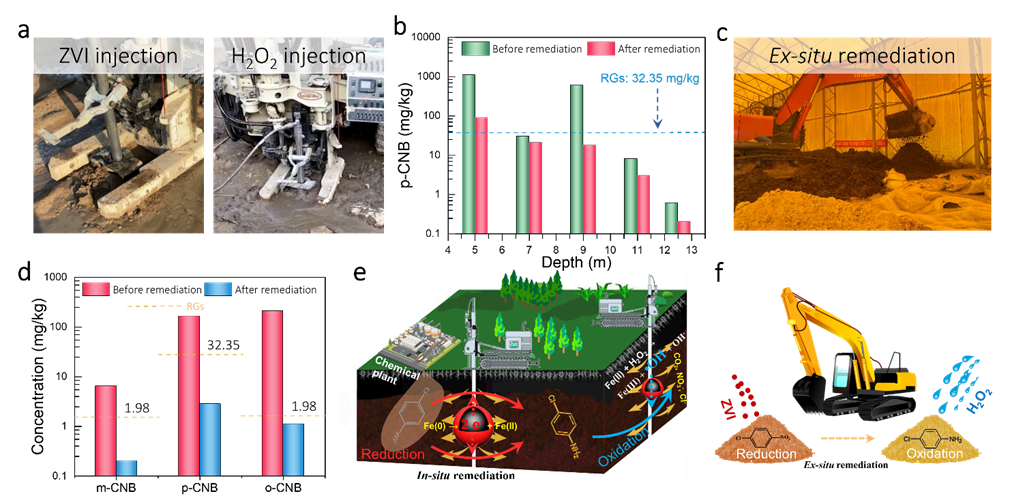
实际土壤中CNBs修复
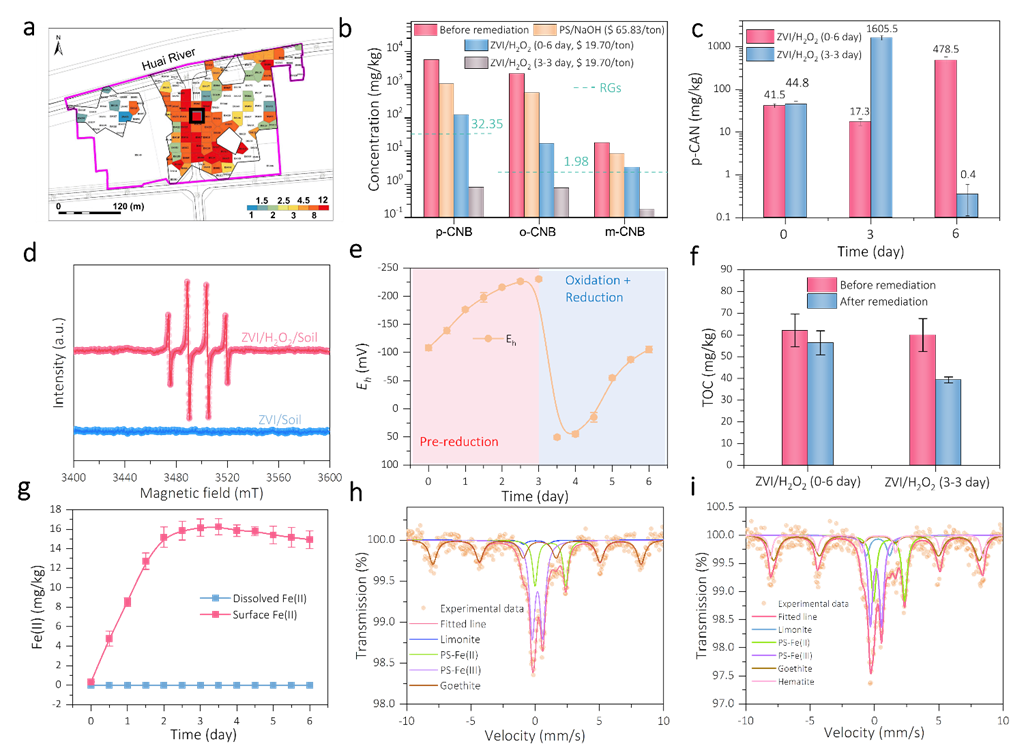
Figure 4. The remediation performance of controllable ROC technology for CNBs in real soil. (a) The contaminated area distribution in remediationsite. (b) The remediation results of collected soil in lab by ZVI/H2O2 based ROC technology and PS/NaOH oxidation technology. (c) Temporal p-CAN concentration in ZVI/H2O2 (0-6 day) and ZVI/H2O2 (3-3 day) processes. (d) The EPR spectra of ZVI/soil and ZVI/H2O2/soil processes with DMPO as spintrapping agent. (e) Time profiles of redox potential (Eh) in ZVI/H2O2 (3-3 day) process. (f) TOC removal performance of ZVI/H2O2 (0-6 day) and ZVI/H2O2 (3-3 day) processes. (g) Time profiles of surface and dissolved Fe(II) concentrations in ZVI/soil process. Mössbauer spectra of the soils (h) before and (i) after ZVI/H2O2 (3-3 day) process. Copyright 2022, Elsevier Inc.
可控的ROC过程(3天还原-3天氧化)可以将实际土壤中的CNBs修复达标,其活性远高于未控制的ROC过程(0天还原-6天氧化)和过硫酸盐/NaOH氧化过程。在修复土壤中CNBs污染时,ZVI依然会将CNBs还原为对应的CANs,并伴随着ZVI表面Fe(II)积累。在加入H2O2后,Fe(II)活化H2O2生成的•OH将CANs矿化。其中,Fe(II)浓度可以作为H2O2加入时间的指示剂。最终,ZVI在土壤中转化为天然铁基矿物,不会引入二次污染。
实际CNBs污染土壤场地修复示范

张礼知 教授,国家杰出青年科学基金获得者,科技部中青年科技创新领军人才计划,教育部长江学者特聘教授,中组部万人计划科技创新领军人才,湖北省高层次人才工程第一层次入选者,湖北省高端人才引领培养计划第二层次入选者,教育部新世纪优秀人才支持计划入选者,洪堡学者(2006-2007,德国马普胶体与界面所)。已获授权专利 30 余项。在 Chem、Nature Communications、Journal of the American Chemical Society、Angewandte Chemie International Edition、Advanced Materials、Environmental Science & Technology 等学术期刊发表论文 290多篇,其中27篇入选 ESI 高被引论文。截至2021年12月论文已被引用 >29200次,H因子103。2008年获得湖北省自然科学二等奖(第一完成人),2011年获湖北省青年科技奖,并入选湖北省自主创新“双百计划”,2012年入选湖北省高端人才引领培养计划和湖北省高层次人才工程,2014年起连续入选 Elsevier发布“化学领域中国高被引学者榜单”,2015年获教育部高等学校科学研究优秀成果奖(科学技术)自然科学二等奖(第一完成人),2018年起连续入选 Clarivate(Web of Science)交叉领域全球高被引科学家榜单,2019 年获湖北省自然科学一等奖(第一完成人)。

艾智慧 教授,湖北省杰出青年,三级教授,博士生导师。主要研究领域为环境化学、环境材料设计合成、污染控制技术。在铁环境化学、高级氧化技术和污染控制领域深耕多年,主持基金委区域创新联合基金重点项目、科技部重点研发项目和国家自然科学基金面上项目多项,作为第二完成人分别荣获2015年度教育部自然科学二等奖和2019年度湖北省自然科学一等奖;2018年进入英国皇家化学会“Top1%高被引中国作者”(环境工程类)榜单,自2019 年连续三年入选 Elsevier 发布中国高被引学者榜单。获授权发明专利十余项;在Angewandte Chemie International Edition、Environmental Science & Technology、Water Research等期刊发表论文110多篇,截至2021年12月论文已被他引9000多次,H因子58。

魏凯,华中师范大学化学学院博士研究生。研究方向为环境污染控制,以第一作者在Advanced Functional Materials、Water research、Applied Catalysis B: Environmental、Chinese Journal of Catalysis和Chemical Engineering Journal Advances上各发表论文1篇。
课题组网站:http://www.irongroup.cn/
备注:
Permissions for reuse of all Figures have been obtained from the original publisher. Copyright 2022, Elsevier Inc.
参考文献:
KaiWei, Yanyan Wan, Minzi Liao, Shiyu Cao, Hao Zhang, Xing Peng, Huayu Gu, CancanLing, Meiqi Li, Yanbiao Shi, Zhihui Ai,* Jingming Gong, and Lizhi Zhang*. A Controllable Reduction-Oxidation Coupling Process for Chloronitrobenzenes Remediation: From Lab to Field Trial. Water Res. 2022, 118453. DOI:10.1016/j.watres.2022.1184538
原文链接:https://www.sciencedirect.com/science/article/abs/pii/S0043135422004079#!